
- How To Calculate Keq Organic Chemistry
- Calculate Keq For The Reaction
- Keq Equation
- What Is The Equilibrium Constant
Calculate the value of Keqat 25.C
F2(g) + 2I- 2F-(aq) + I2(g) Tekla 20.1 crack free download.
I'm thinking the fact that it's at equilibrium is what I'm not accounting for, but here is what I've done.
May 17, 2009 Calculate the Keq for the following reaction: N2O4(g) 2NO2(g) If N2O2 = 0.0185 mol/L and NO2 = 0.0627 mol/L. If the way I wrote the problem looks confusing can someone just tell me how to calculate Keq and maybe I can figure out the rest. Thank you for your answers in advance. Find Kb for ammonia: This would simply be the given Keq for the reaction. Kb is just the Keq of a specific type of reaction between a base and water. Ammonia is a weak base. Find Ka of conjugate acid. This is would equal the Keq of the reverse reaction. The Keq of the reverse reaction is the reciprocal of the the forward reaction.
For Ecell I have F2 as my cathode and I as my anode.
I got 2.331 for this.
I got 2.331 for this.
Next I got my G value. G= -nFE -(2)(9.65x 104)(2.331)
I got -449,883 J for this.(I left it in joules for the next part)
I got -449,883 J for this.(I left it in joules for the next part)
Wilcom embroidery studio e3 dongle cracker. Finally, I used the rearranged equation for the relation between G and K. Sky go crack serial keygen torrent.
Keq= ex of -(G/RT) -(-449,883/8.314 x 298)=181.58
e181.58= The wrong answer. Tekken 6 mobile game free download.
Any thoughts? Thanks
Great Thanks to Tina Nye (A-5; 05-06) for much work on graphics: making a rough idea reality!
Chemical equilibrium
A system in equilibrium is like our ants up there! As long as the ants work at the same speed, the piles of sand remain in equilibrium. Neither gets bigger or smaller (It is important to note however that the piles are not the same size!). The work that each ant does exactly offsets what the other is doing. Products are being stacked up and the same rate that they are being taken away. Reactants are being stacked up at exactly the same rate that they are being taken away. If this were a chemical reaction, reactants would be colliding to make product at the same rate that products would be colliding with each other to make fresh reactant again! The reaction never really stops, but the forward reaction and reverse reaction proceed and the same rate, so we don't see a change in the size of the piles! Nifty Huh!!!!?
Chemical equilibrium is reached when the rates for the forward and reverse chemical reactions are equal for a chemical system. Or you could say when products are being made as fast as they are breaking down to form reactants again. The concentrations of products and reactants are generally NOT EQUAL.
To know what the concentrations are, you use the Equilibrium Constant expression (Keq).
How To Calculate Keq Organic Chemistry
Keq = [products]/[reactants]. Its a ratio : ) So Keq > 1 favors products, Keq < 1 favor reactants. Adobe photoshop 7.0 free for mac.
For a general equation like:
Calculate Keq For The Reaction
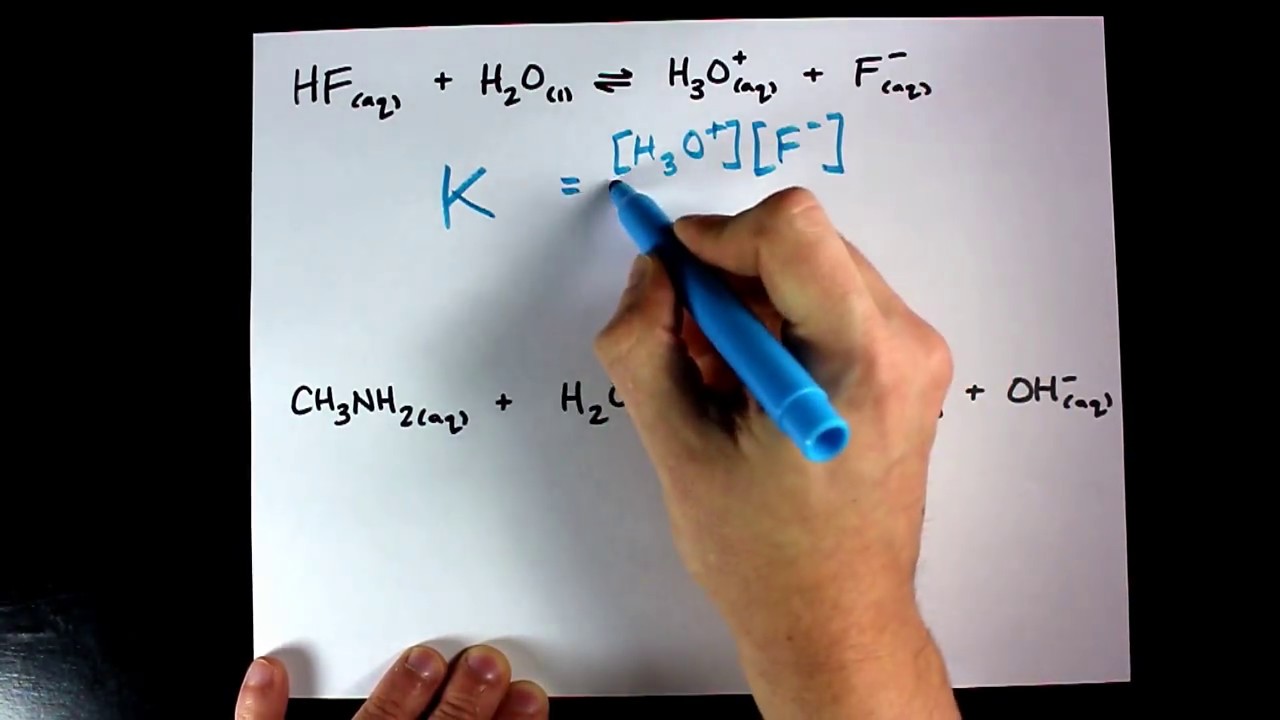
Asus vw246h driver for mac. wA + xB <> yC + zD
Keq Equation
Remember, the square brackets mean concentration, in molarity, if it is given in some other unit you must calculate molarity. Coefficients in the chemical equation become exponents in the Keq expression. Leave out any solids or pure liquids since they have undefined molarities ; ).
Keq= If you spend time with the following notes, you'll master this topic in no time. Midnight sun stephenie meyer pdf torrent.
What Is The Equilibrium Constant
Predicting final concentrations and cheating while were doing it.
